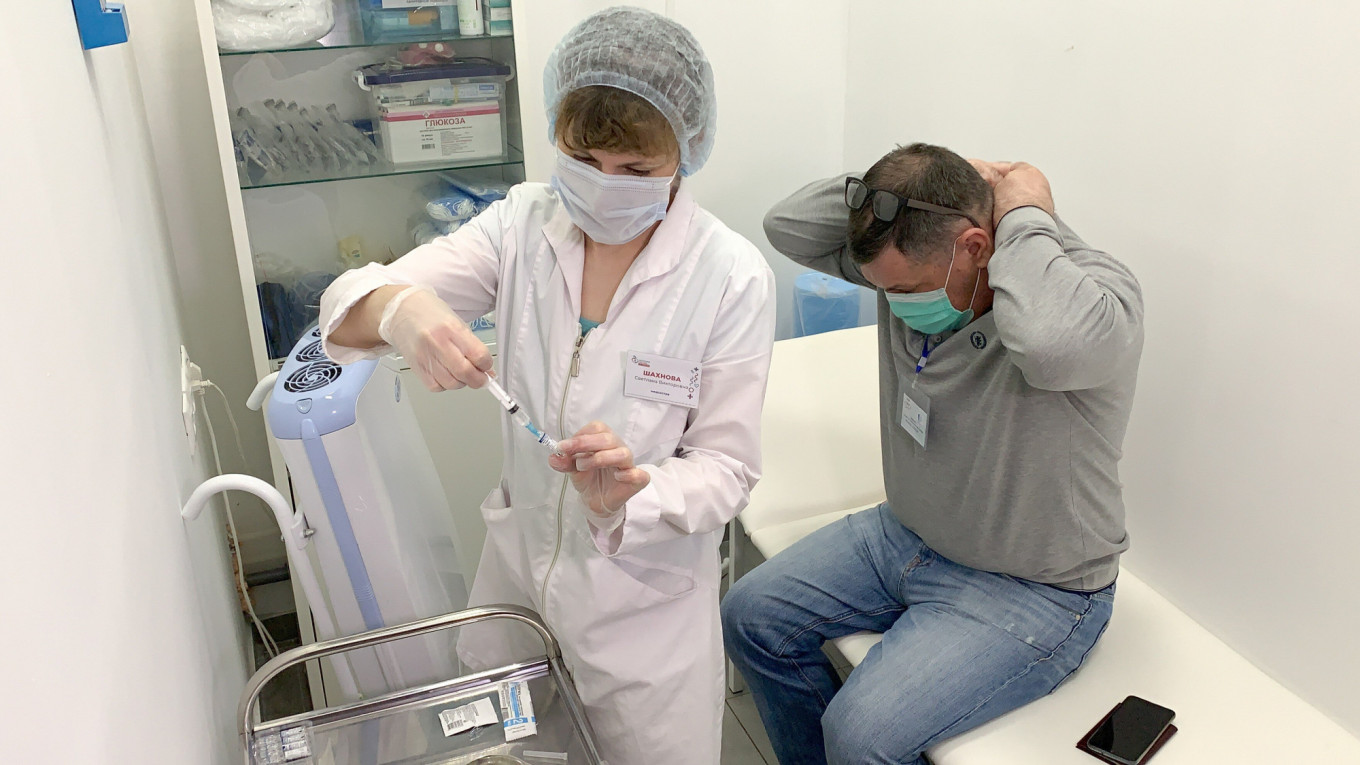
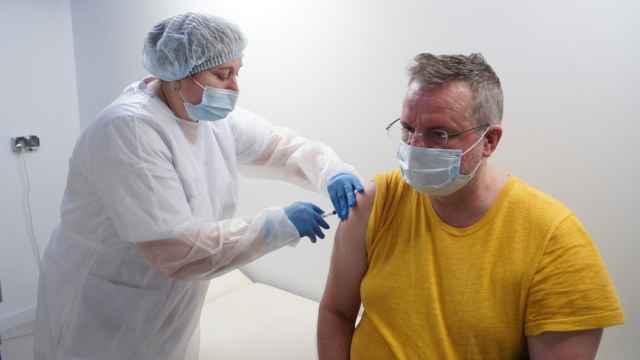
The developer of Russia's Sputnik V coronavirus vaccine said Wednesday its jab did not cause blood clots, a potential side effect that has disrupted rollouts in several Western countries.
The United States has recommended pausing its rollout of the Johnson & Johnson vaccine after one person out of nearly 7 million Americans died from a rare type of clot in the brain following inoculation.
Denmark on Wednesday became the first country in Europe to suspend use of the AstraZeneca jab after reports of rare but serious cases of blood clots among those that had received the vaccine.
Like the Johnson & Johnson and AstraZeneca vaccines, Russia's Sputnik V jab uses adapted strains of the adenovirus that causes the common cold.
On Wednesday, the state-run Gamaleya research institute, which developed Sputnik V vaccine, said there was no risk of blood clots from its jab.
"A comprehensive analysis of adverse events during clinical trials and over the course of mass vaccinations with the Sputnik V vaccine showed that there were no cases of cerebral venous sinus thrombosis," it said in a statement.
Russia's registration of Sputnik in August last year triggered criticism both at home and abroad over the fast-track procedure, but a leading medical journal said this year it was safe and highly effective.
The vaccine has been registered for use in dozens of countries.
The Gamaleya institute said it was "ready to share its purification technology with other vaccine producers in order to help them minimize the risk of adverse effects during vaccination."
President Vladimir Putin said Wednesday he had received the second dose of a vaccine against the coronavirus and encouraged Russians to follow his example.
He has not said which of Russia's three vaccines — EpicVacCorona and CoviVac in addition to Sputnik V — he received.
A Message from The Moscow Times:
Dear readers,
We are facing unprecedented challenges. Russia's Prosecutor General's Office has designated The Moscow Times as an "undesirable" organization, criminalizing our work and putting our staff at risk of prosecution. This follows our earlier unjust labeling as a "foreign agent."
These actions are direct attempts to silence independent journalism in Russia. The authorities claim our work "discredits the decisions of the Russian leadership." We see things differently: we strive to provide accurate, unbiased reporting on Russia.
We, the journalists of The Moscow Times, refuse to be silenced. But to continue our work, we need your help.
Your support, no matter how small, makes a world of difference. If you can, please support us monthly starting from just $2. It's quick to set up, and every contribution makes a significant impact.
By supporting The Moscow Times, you're defending open, independent journalism in the face of repression. Thank you for standing with us.
Remind me later.