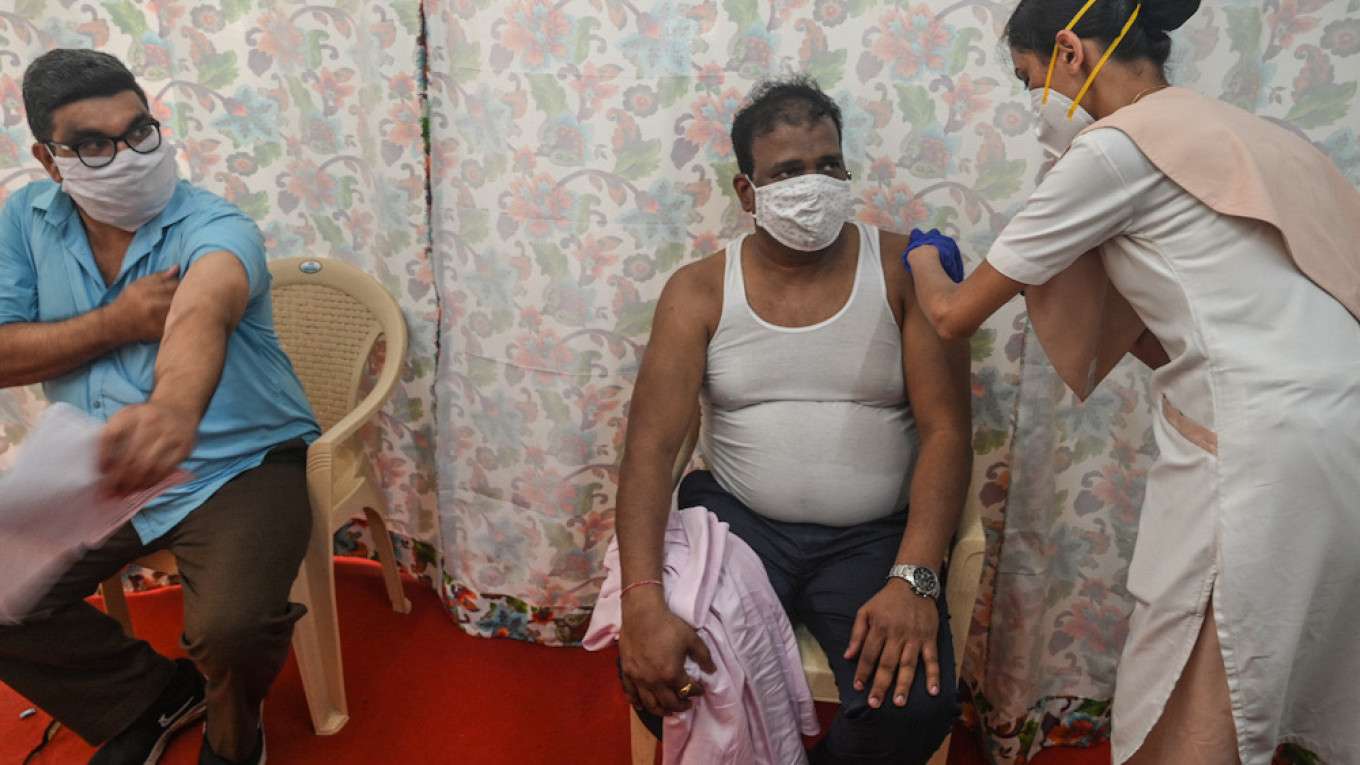
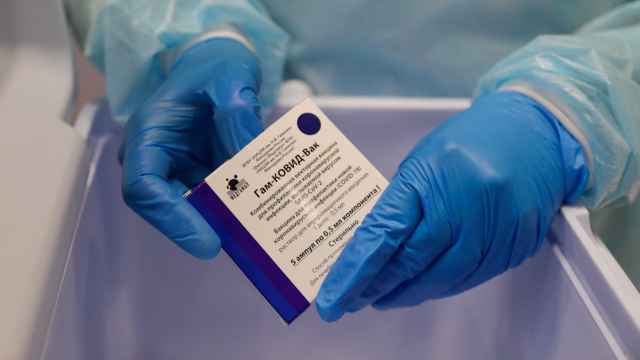
India has approved Russia’s Sputnik V coronavirus vaccine in an effort to boost the country’s vaccination drive amid soaring case numbers, the Russian Direct Investment Fund (RDIF) that markets the jab said Tuesday.
While India has already approved two other vaccines, the Oxford-AstraZeneca shot and the India-developed Covaxin, the country of 1.3 billion people continues to battle the world’s second-highest number of new coronavirus infections. India is the 60th country in the world to approve Sputnik V, according to the RDIF.
India’s Drug Controller General authorized Sputnik V for emergency use based on the jab’s clinical trial results in Russia and local Phase 3 trial results conducted in partnership with pharmaceutical company Dr. Reddy’s Laboratories, the RDIF said.
“We appreciate the decision of India’s regulatory bodies to grant authorization for Sputnik V,” RDIF head Kirill Dmitriev said in a statement.
“Approval of the vaccine is a major milestone as Russia and India have been developing an extensive cooperation on clinical trials of Sputnik V in India and its local production,” he said.
Dmitriev told Indian broadcaster NDTV that the first doses could be ready by late April or May, with ramped up production by June.
"We believe by June, we will really be at good production capacity in India and will become a very meaningful player in vaccination program in India," he added.
Sputnik V already has production agreements in India, home to the world's biggest vaccine manufacturer, to produce 852 million doses.
AFP contributed reporting.
A Message from The Moscow Times:
Dear readers,
We are facing unprecedented challenges. Russia's Prosecutor General's Office has designated The Moscow Times as an "undesirable" organization, criminalizing our work and putting our staff at risk of prosecution. This follows our earlier unjust labeling as a "foreign agent."
These actions are direct attempts to silence independent journalism in Russia. The authorities claim our work "discredits the decisions of the Russian leadership." We see things differently: we strive to provide accurate, unbiased reporting on Russia.
We, the journalists of The Moscow Times, refuse to be silenced. But to continue our work, we need your help.
Your support, no matter how small, makes a world of difference. If you can, please support us monthly starting from just $2. It's quick to set up, and every contribution makes a significant impact.
By supporting The Moscow Times, you're defending open, independent journalism in the face of repression. Thank you for standing with us.
Remind me later.