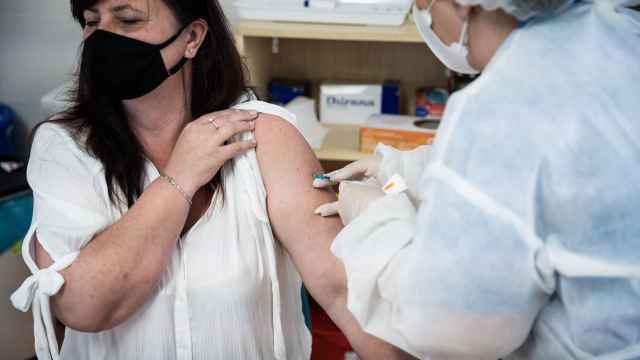
Russia has approved clinical trials of a combination of its Sputnik V and the Anglo-Swedish drugmaker AstraZeneca’s coronavirus vaccine that will run through next spring.
Five Russian clinics will hold AstraZeneca and Sputnik V trials involving 150 people, with testing wrapping up in early March 2022, according to the entry in Russia's state drug register published Monday.
The Sputnik V and AstraZeneca vaccines are both two-component adenovirus vector vaccines. Sputnik V's two doses use two different viral vectors.
AstraZeneca had announced plans last December to partner with Sputnik V's developer in combining the two adenovirus-based vaccines, saying it “may prove important for revaccinations.”
Sputnik V's developers believe that its effectiveness against the virus, including highly contagious new mutations, can be increased by combining it with other vaccines.
But the Russian Health Ministry’s ethics committee did not grant approval for Phase Two and Phase Three clinical trials of the Sputnik V/AstraZeneca combination in late May.
Sputnik V’s global marketer, the Russian Direct Investment Fund (RDIF), welcomed the approval of the joint clinical trials, saying similar “vaccine cocktail” trials have been ongoing in the UAE, Argentina and Azerbaijan since February.
The AstraZeneca-Oxford University vaccine, known as AZD2816, is seen as a cheaper and more mobile alternative to its rivals due to its ability to be transported at normal fridge temperatures.
The AstraZeneca vaccine’s overall efficacy at preventing people from developing Covid-19 symptoms after two doses was assessed at 70% compared to Sputnik V’s 91.6%.
A Message from The Moscow Times:
Dear readers,
We are facing unprecedented challenges. Russia's Prosecutor General's Office has designated The Moscow Times as an "undesirable" organization, criminalizing our work and putting our staff at risk of prosecution. This follows our earlier unjust labeling as a "foreign agent."
These actions are direct attempts to silence independent journalism in Russia. The authorities claim our work "discredits the decisions of the Russian leadership." We see things differently: we strive to provide accurate, unbiased reporting on Russia.
We, the journalists of The Moscow Times, refuse to be silenced. But to continue our work, we need your help.
Your support, no matter how small, makes a world of difference. If you can, please support us monthly starting from just $2. It's quick to set up, and every contribution makes a significant impact.
By supporting The Moscow Times, you're defending open, independent journalism in the face of repression. Thank you for standing with us.
Remind me later.